Healthcare · Example application
Clinical Research
Clinical trial operations, enrollment, adverse events & regulatory
About this example
Clinical Research: an example of what's possible on the Kinetic Platform
Clinical Research is one of 48+ working applications in Kinetic AI Labs — a portfolio of example apps built on the Kinetic Platform to show how quickly real, production-grade software can be assembled when the platform handles workflow execution, data, identity, and integrations. These applications are not commercial products. They are reference implementations: live demonstrations of healthcare use cases that customers and partners can study, fork, or use as starting points for their own builds.
What this application does
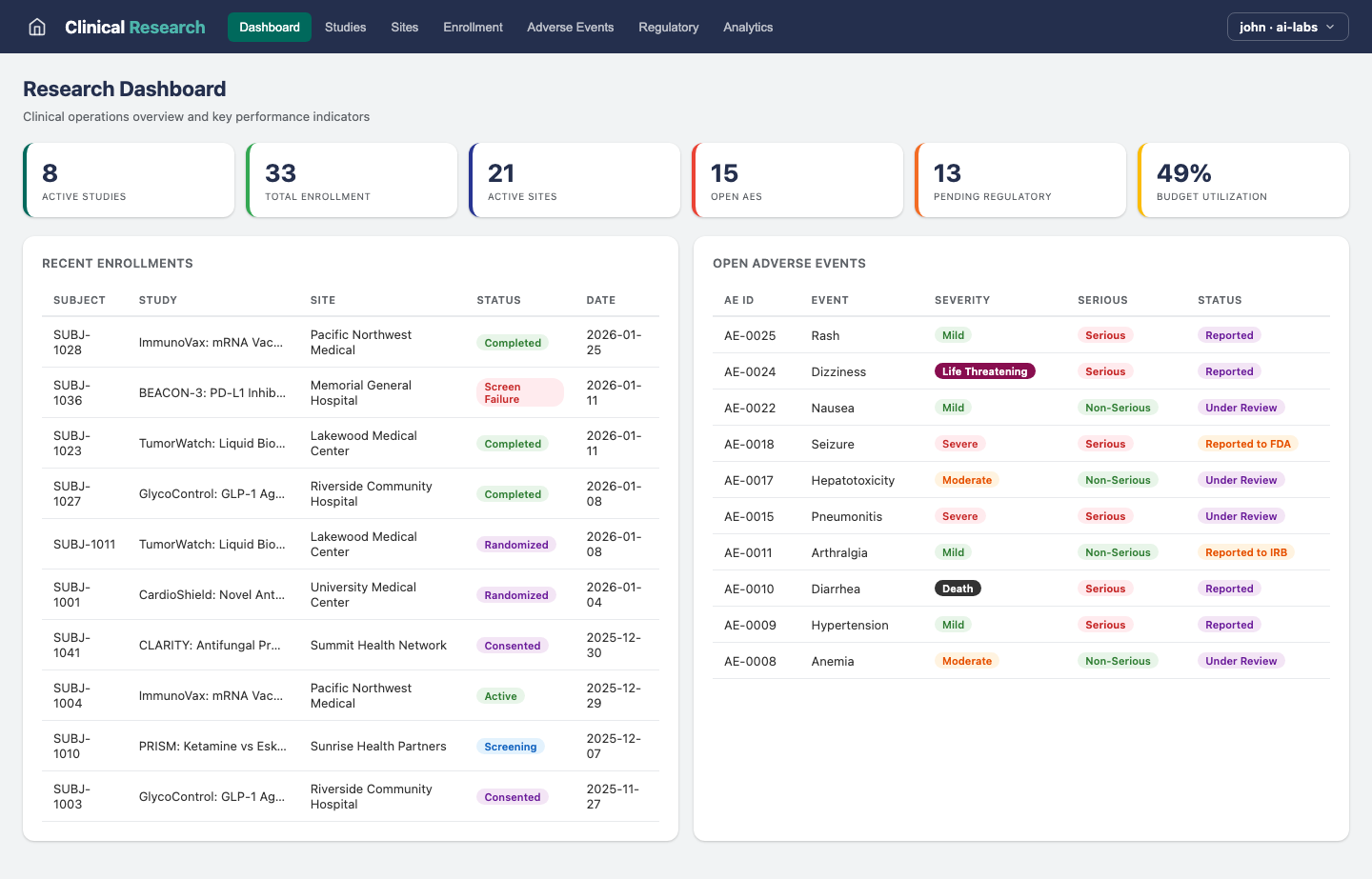
End-to-end clinical research management covering studies, multi-site coordination, patient enrollment, adverse event reporting, regulatory filings, and analytics. Dashboard shows active studies, enrollment progress, and regulatory deadlines.
Application modules
Key capabilities
What Clinical Research demonstrates about building on the Kinetic Platform.
Study Management
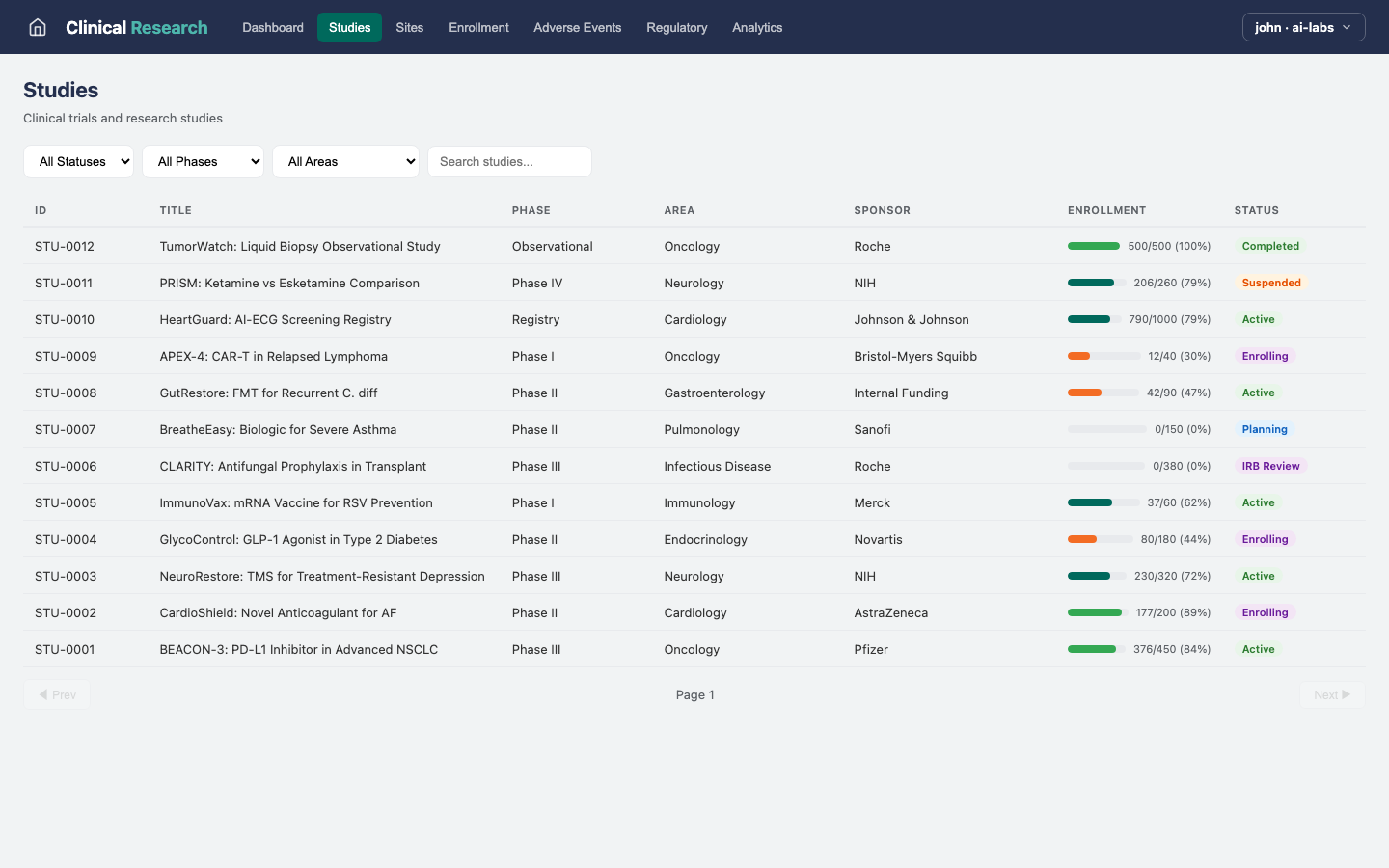
Define and track research studies with protocols, milestones, and site assignments.
Enrollment Tracking
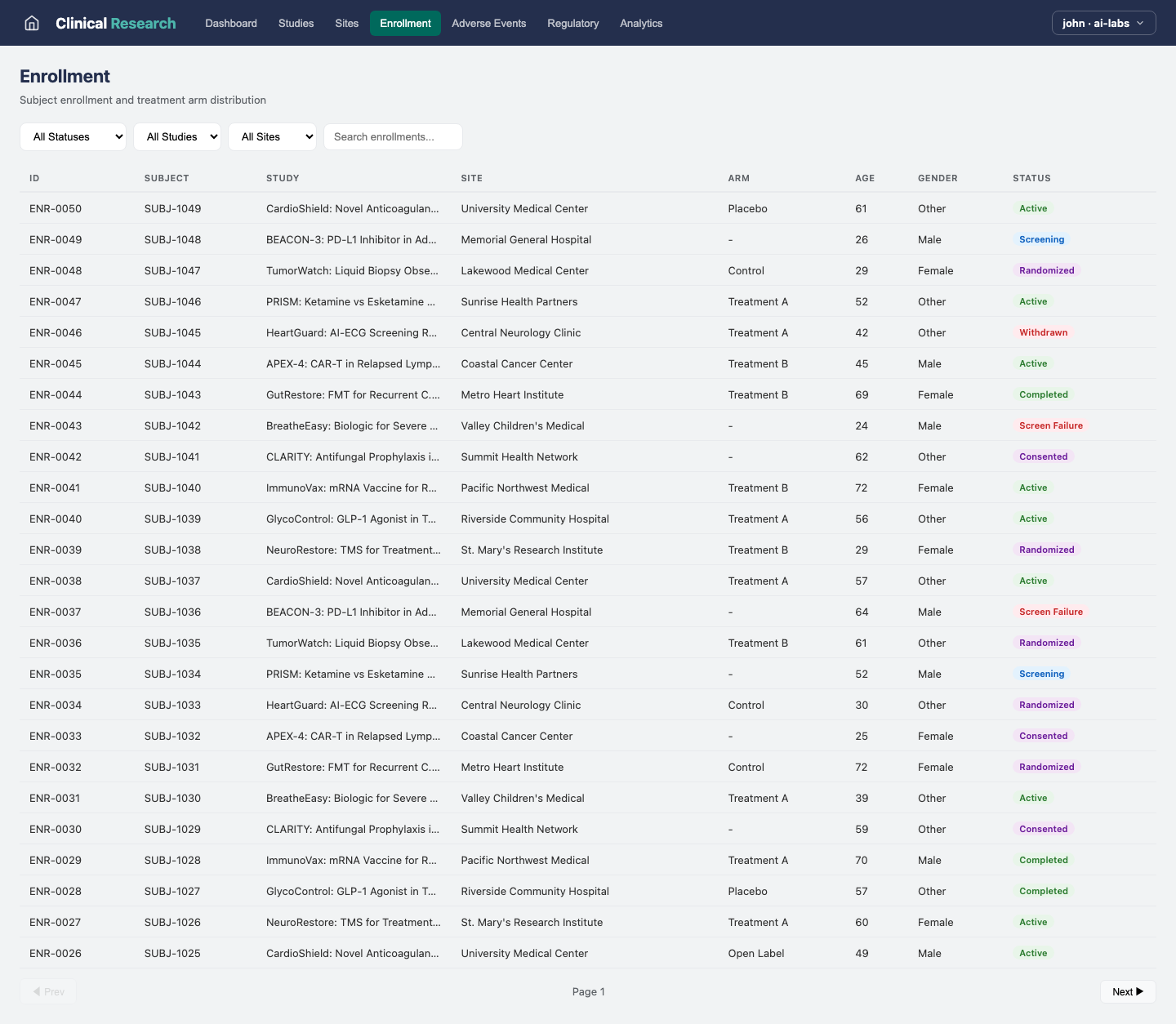
Monitor patient enrollment across sites with real-time progress against targets.
Adverse Events
Structured capture and classification of adverse events with regulatory notification.
Regulatory Compliance
Track regulatory submissions, approvals, and compliance across jurisdictions.
Screenshots
See Clinical Research in action
These screenshots are captured directly from the live AI Labs instance — every screen below renders against real workflows and data running on the Kinetic Platform.
Dashboard
Studies
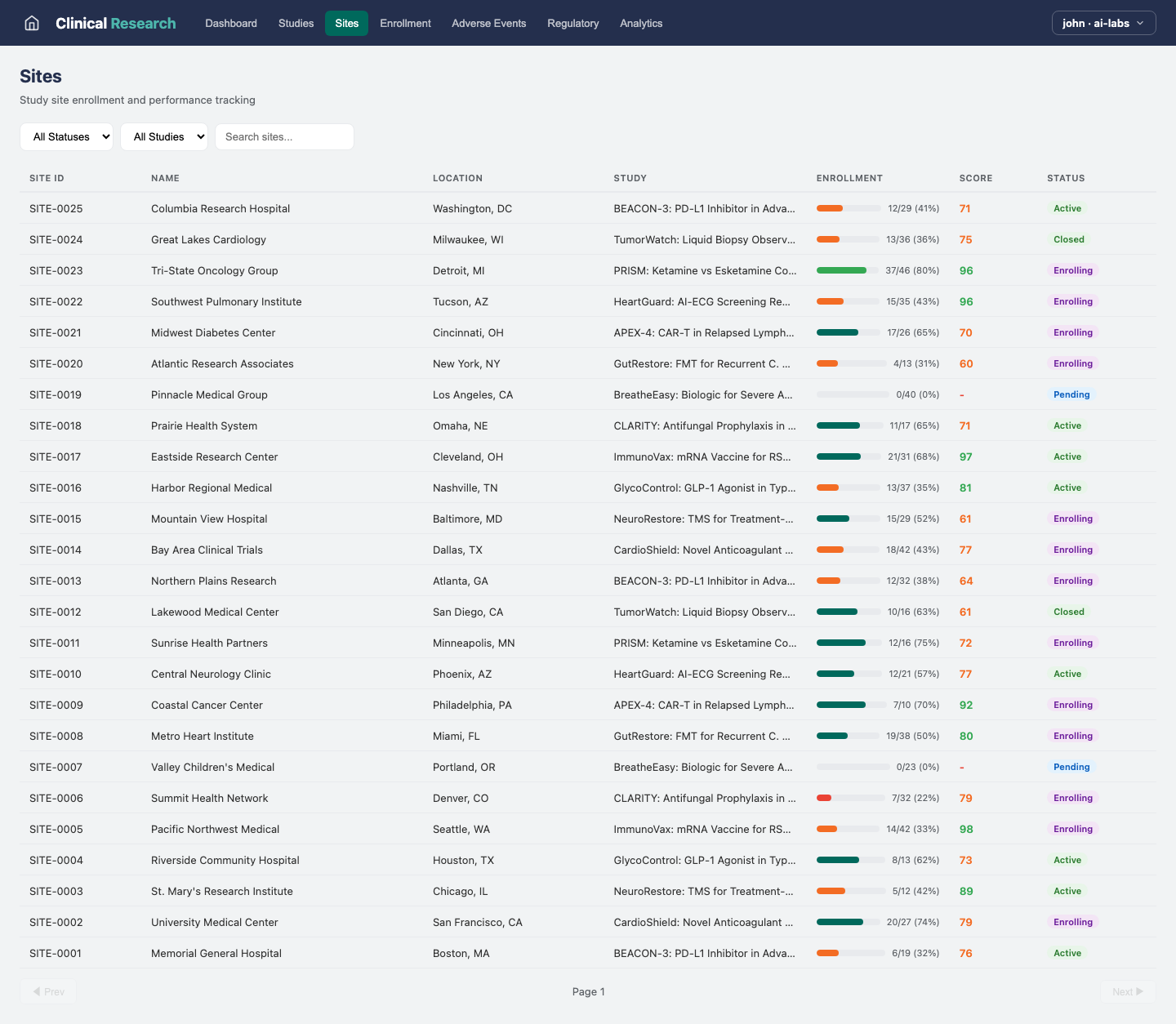
Sites
Enrollment
Adverse Events
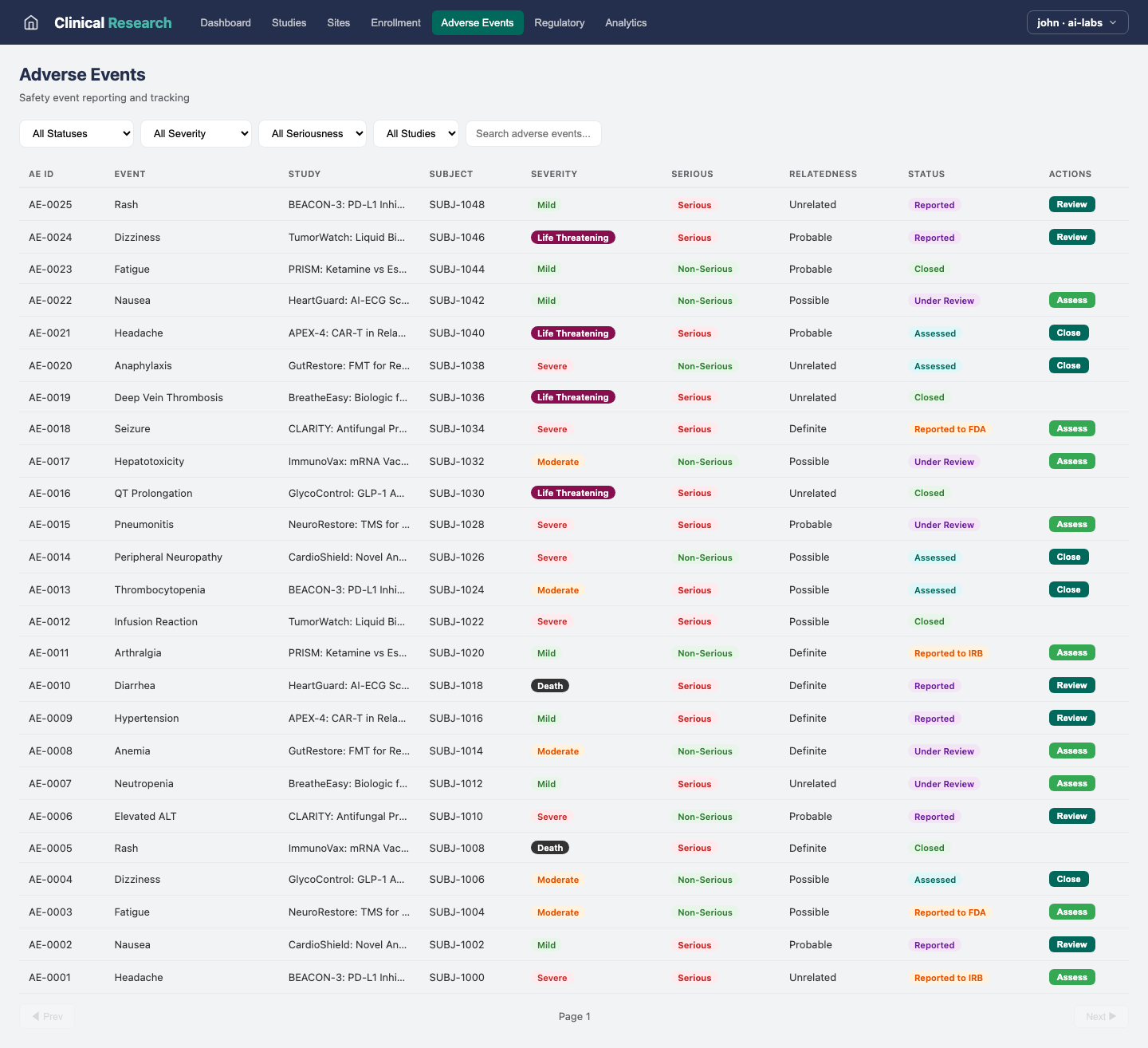
Regulatory
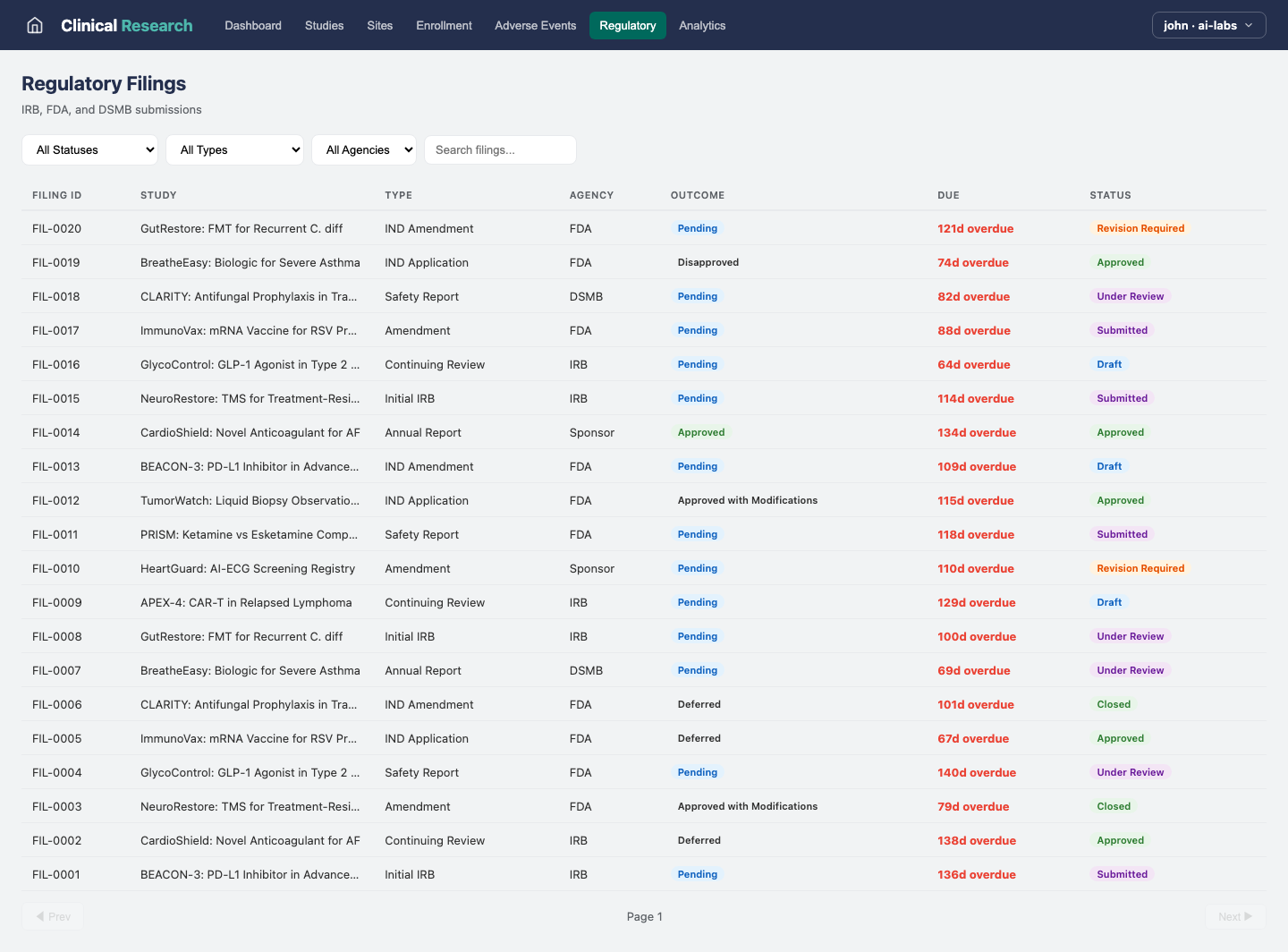
Analytics
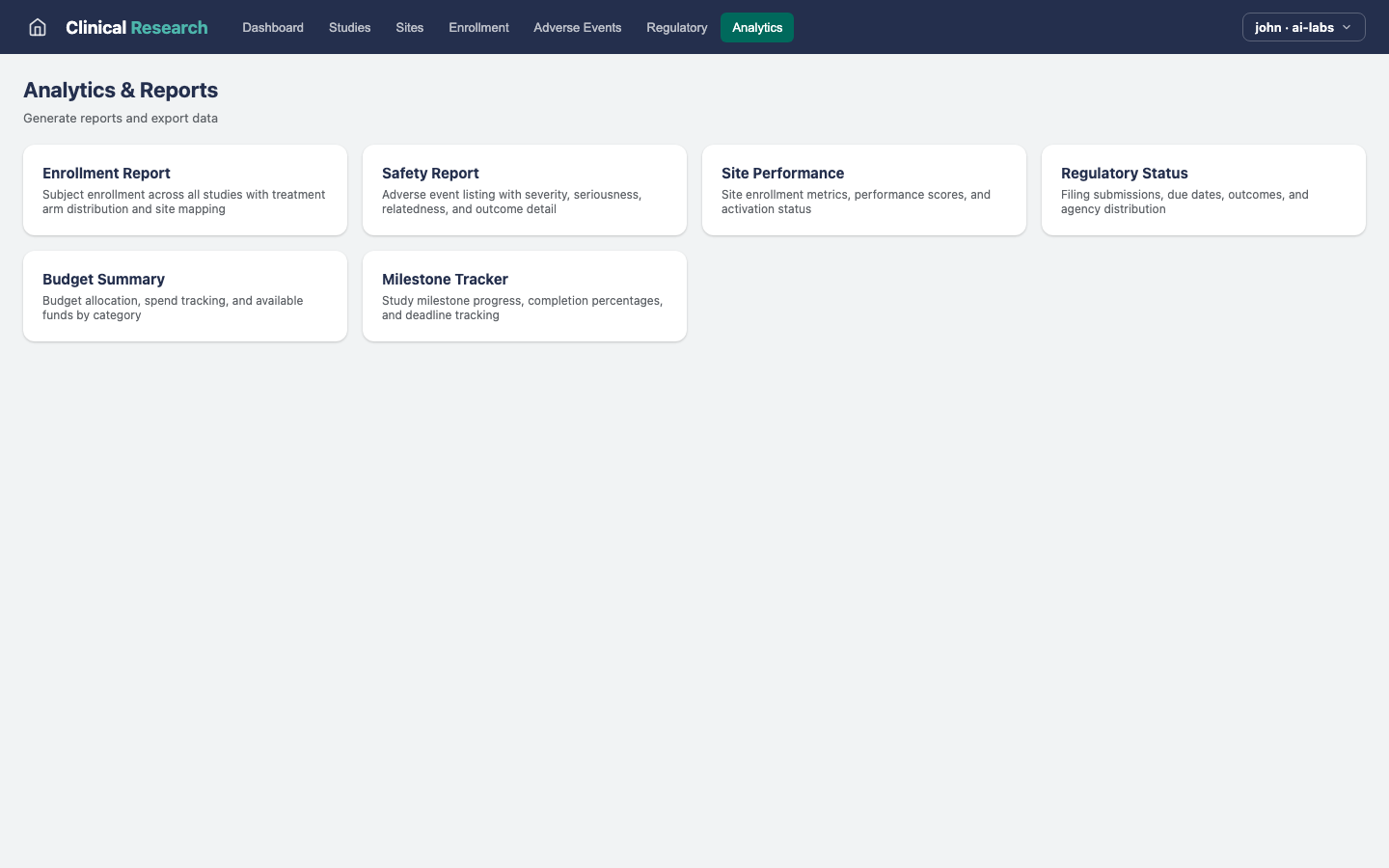
What this example demonstrates
Why Clinical Research matters as a reference implementation
Clinical Research shows how a healthcare application can be built on a workflow orchestration layer instead of a custom-coded stack. The screens you see above are not mockups — they are full Kinetic forms, tables, dashboards, and views, wired into deterministic workflows that run the underlying business logic.
Because every example in AI Labs runs on the same Kinetic Platform instance, they share one workflow engine, one identity model, one integration layer, and one audit trail. That means the patterns proven in Clinical Research — study management, enrollment tracking, and the rest of its capabilities — transfer directly into customer environments without re-platforming.
Built on Kinetic
Same platform, every application
Clinical Research runs on the same Kinetic Platform instance as every other AI Labs example. Same workflow engine. Same security model. Same integration layer. Clinical Research is a working illustration of how quickly a production-ready application can be built when the platform handles the hard parts — workflow execution, data management, role-based access, and audit trails.
Want to see Clinical Research live?
Schedule a demo and we'll walk through Clinical Research on the Kinetic Platform — or talk about adapting it (or building a new one) for your environment.